
DEXTENZA is a corticosteroid indicated for:1
- The treatment of ocular inflammation and pain following ophthalmic surgery.
- The treatment of ocular itching associated with allergic conjunctivitis.
Please see Important Safety Information below.
DEXTENZA provides an opportunity for physicians to alleviate their patients of the burden of administration of their post-op steroid drop regimen.2 One of our innovative inserts can replace the need for patients to administer ~70 steroid eye drops.3
DEXTENZA is a preservative-free intracanalicular insert that is inserted in the lower lacrimal punctum, a natural opening in the eye lid, and into the canaliculus.1 DEXTENZA is designed to deliver a tapered dose of steroid (dexamethasone) to the ocular surface for up to 30 days.1,4 Following treatment, DEXTENZA resorbs and exits the nasolacrimal system without the need for removal.1

INSERTION OF DEXTENZA
Video courtesy of Dr. Joe Gira.
DEXTENZA is dexamethasone delivered via hydrogel technology1,2

ACTIVATES1,2
- With moisture
- Swells to fit comfortably and securely in the canaliculus
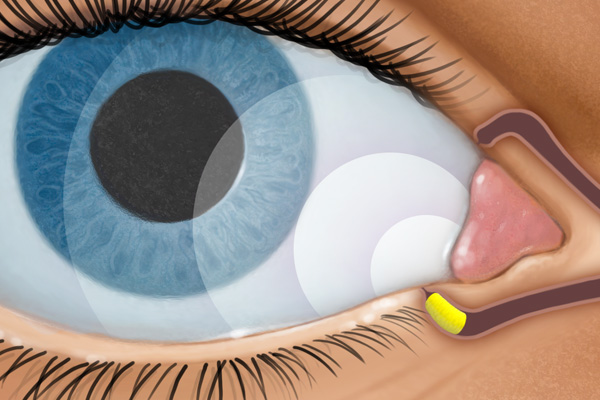
RELEASES1,2
- Dexamethasone for up to 30 days
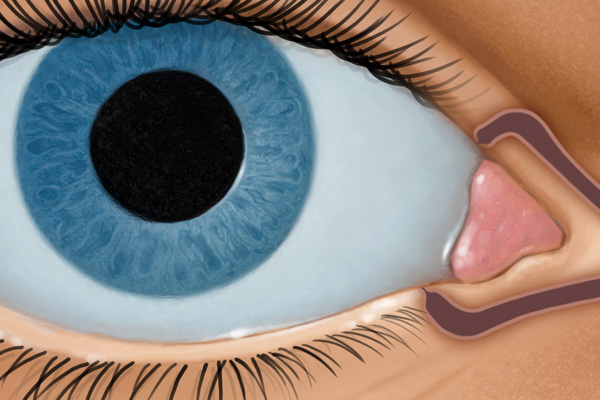
RESORBS1,2
- Slowly through the course of treatment
- Clears via the nasolacrimal duct
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
DEXTENZA is contraindicated in patients with active corneal, conjunctival or canalicular infections, including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella; mycobacterial infections; fungal diseases of the eye, and dacryocystitis.
WARNINGS AND PRECAUTIONS
Intraocular Pressure Increase – Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. Steroids should be used with caution in the presence of glaucoma. Intraocular pressure should be monitored during treatment.
Bacterial Infections – Corticosteroids may suppress the host response and thus increase the hazard for secondary ocular infections. In acute purulent conditions, steroids may mask infection and enhance existing infection.
Viral Infections – Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex).
Fungal Infections – Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use. Fungal culture should be taken when appropriate.
Delayed Healing – Use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation.
Other Potential Corticosteroid Complications – The initial prescription and renewal of the medication order of DEXTENZA should be made by a physician only after examination of the patient with the aid of magnification, such as slit lamp biomicroscopy, and, where appropriate, fluorescein staining. If signs and symptoms fail to improve after 2 days, the patient should be re-evaluated.
ADVERSE REACTIONS
Ocular Inflammation and Pain Following Ophthalmic Surgery
The most common ocular adverse reactions that occurred in patients treated with DEXTENZA were: anterior chamber inflammation including iritis and iridocyclitis (10%), intraocular pressure increased (6%), visual acuity reduced (2%), cystoid macular edema (1%), corneal edema (1%), eye pain (1%), and conjunctival hyperemia (1%). The most common non-ocular adverse reaction was headache (1%).
Itching Associated with Allergic Conjunctivitis
The most common ocular adverse reactions that occurred in patients treated with DEXTENZA were: intraocular pressure increased (3%), lacrimation increased (1%), eye discharge (1%), and visual acuity reduced (1%). The most common non-ocular adverse reaction was headache (1%).
Please see full Prescribing Information.
REFERENCES: 1. DEXTENZA [prescribing information]. Bedford, MA: Ocular Therapeutix, Inc; 2021. 2. Walters T, et al. J Clin Exp Ophthalmol. 2016;7(4):1-11. 3. Data on file 00663. Ocular Therapeutix, Inc. 4. Sawhney AS, et al. US patent 8,409,606 B2. April 2, 2013.
